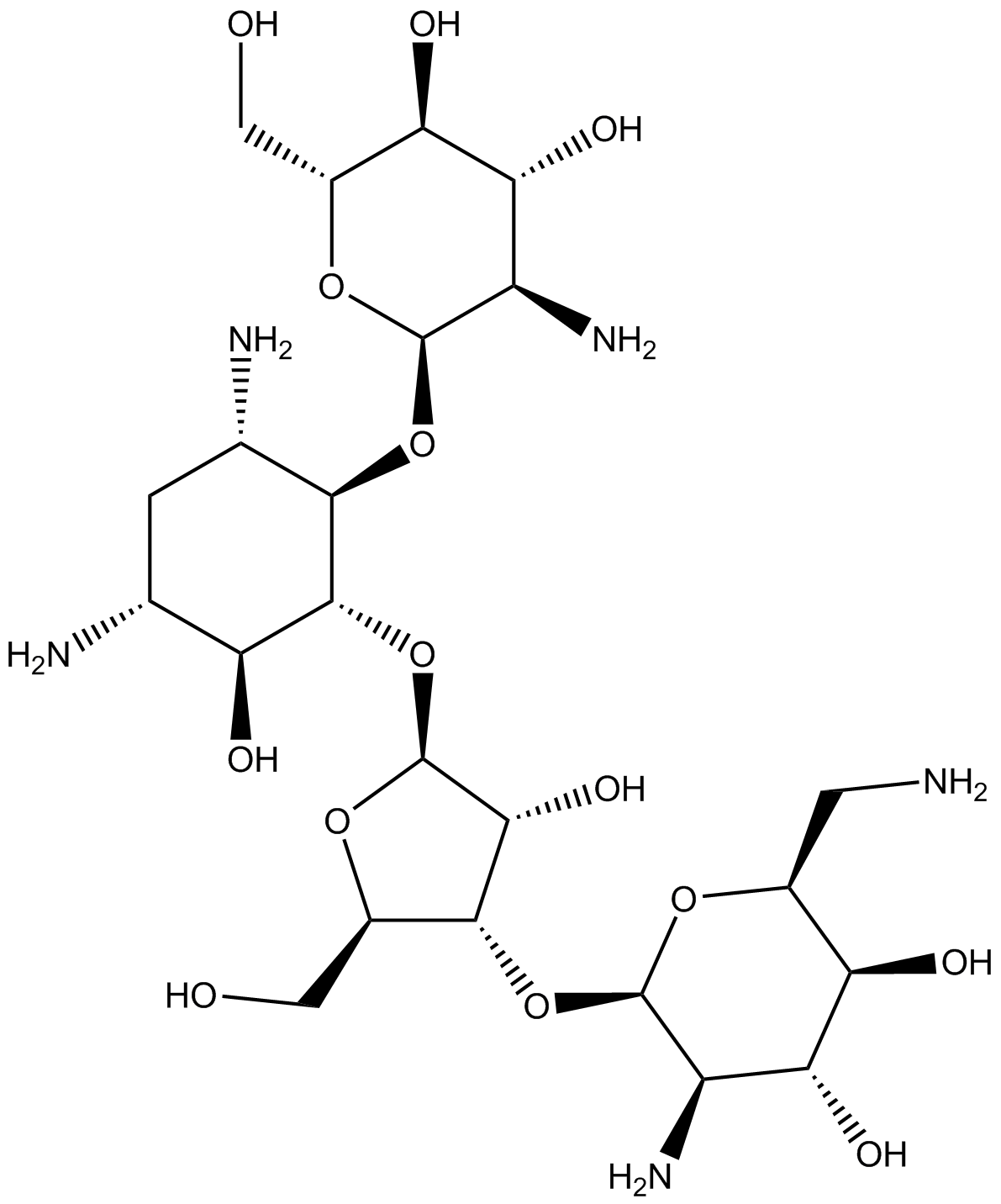
Paromomycin Sulfate
| ||||||||||||||||||||||||||||||||||||||||||||||
| Solubility | Water |
| Physical Form | Solid |
| Storage Temp. | Room Temperature |
| UPC / SKU | P710 |
| CAS NUMBER | 1263-89-4 |
| Formula Weight | 713.7 |
| Formula | C23H45N5O14 • H2SO4 |
| Synonyms | O-2-Amino-2-deoxy-α-D-glucopyranosyl-(1-4)-O-[O-2,6-diamino-2,6-dideoxy-β-L-idopyranosyl-(1-3)-β-D-ribofuranosyl-(1-5)]-2-deoxy-D-streptamine Sulfate |
| Storage Temp. | Room Temperature |
| Tariff Code | 2941.90.1010 |
| Risk Info (R) | 61-36/37/38 |
| Safety Info (S) | 24/25-36 |
No information available
P710 Paromomycin Sulfate
| Synonyms: | O-2-Amino-2-deoxy-a-D-glucopyranosyl-(1?4)-O-[O-2,6-diamino-2,6-dideoxy-ß-L-idopyranosyl-(1?3)-ß-D-ribofuranosyl-(1?5)]-2-deoxy-D-streptamine Sulfate |
| CAS: | 1263-89-4 |
| Formula: | C23H45N5O14•H2SO4 |
| Mol. Weight: | 713.7 |
| Properties | |
|---|---|
| Form: | Powder |
| Appearance: | Cream to Yellow Powder |
| Application: | Plant Tissue Culture Antibiotic |
| Solubility: | Soluble in Water |
| Storage Temp: | Room Temperature |
| Other Notes: | Plant Tissue Culture Tested |
Application Notes
Paromomycin is an aminoglycoside antibiotic with similar spectrum to that of neomycin. Its mode of action is to inhibit protein synthesis at the initiation and elongation stages by binding to the 16S ribosomal RNA. Paromomycin is effective against various protozoa, e.g., Leishmania spp., Entamoeba histolytica, and Cryptosporidium spp. It has been reported that cross-resistance between paromomycin with other antibiotics, e.g., kanamycin, framycin, neomycin and streptomycin.2
In molecular biology applications, paromomycin has been reported to be used as a selection agent.3, 4
Please Note: While PhytoTechnology LaboratoriesT tests each lot of this product with two or more plant cell/ tissue culture lines, it is the sole responsibility of the purchaser to determine the appropriateness of this product for the specific plants that are being cultured and applications that are being used.
References
- Merck 13, 7113
- Martindale: The Complete Drug Reference, 35th ed., Paul S. Blake, Ed. (Royal Pharmaceutical Society, 2007), p. 759.
- Kimberly A. Torbert, Howard W. Rines, and David A. Somers. 1995. Use of paromomycin as a selective agent for oat transformation. Plant Cell Reports. Vol 14(10). Pp. 635-640.
- César Petri, Sonia López-Noguera, Nuria Alburquerque, José Egea, Lorenzo Burgos. 2008. An antibiotic-based selection strategy to regenerate transformed plants from apricot leaves with high efficiency. Plant Science. Vol 175(6). Pp. 777-783.